our decellurisation timelapse
|
|
We conducted a decellurisation experiment using a DIY protocol. This timelapse video shows the pig heart we used throughout the five hour process.
-Jed |
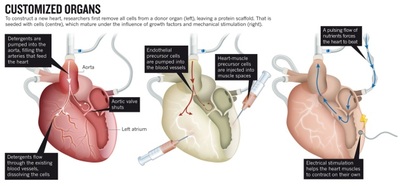
Decellurisation of the heart.
Doris Taylor doesn't take it as an insult when people call her Dr Frankenstein. “It was actually one of the bigger compliments I've gotten,” she says — an affirmation that her research is pushing the boundaries of the possible. Given the nature of her work as director of regenerative medicine research at the Texas Heart Institute in Houston, Taylor has to admit that the comparison is apt. She regularly harvests organs such as hearts and lungs from the newly dead, re-engineers them starting from the cells and attempts to bring them back to life in the hope that they might beat or breathe again in the living.
Taylor is in the vanguard of researchers looking to engineer entire new organs, to enable transplants without the risk of rejection by the recipient's immune system. The strategy is simple enough in principle. First remove all the cells from a dead organ — it does not even have to be from a human — then take the protein scaffold left behind and repopulate it with stem cells immunologically matched to the patient in need. Voilà! The crippling shortage of transplantable organs around the world is solved.
In practice, however, the process is beset with tremendous challenges. Researchers have had some success with growing and transplanting hollow, relatively simple organs such as tracheas and bladders (seego.nature.com/zvuxed). But growing solid organs such as kidneys or lungs means getting dozens of cell types into exactly the right positions, and simultaneously growing complete networks of blood vessels to keep them alive. The new organs must be sterile, able to grow if the patient is young, and at least nominally able to repair themselves. Most importantly, they have to work — ideally, for a lifetime. The heart is the third most needed organ after the kidney and the liver, with a waiting list of about 3,500 in the United States alone, but it poses extra challenges for transplantation and bioengineering. The heart must beat constantly to pump some 7,000 litres of blood per day without a back-up. It has chambers and valves constructed from several different types of specialized muscle cells called cardiomyocytes. And donor hearts are rare, because they are often damaged by disease or resuscitation efforts, so a steady supply of bioengineered organs would be welcome.
Taylor, who led some of the first successful experiments to build rat hearts1, is optimistic about this ultimate challenge in tissue engineering. “I think it's eminently doable,” she says, quickly adding, “I don't think it's simple.” Some colleagues are less optimistic. Paolo Macchiarini, a thoracic surgeon and scientist at the Karolinska Institute in Stockholm, who has transplanted bioengineered tracheas into several patients, says that although tissue engineering could become routine for replacing tubular structures such as windpipes, arteries and oesophagi, he is “not confident that this will happen with more complex organs”.
Yet the effort may be worthwhile even if it fails, says Alejandro Soto-Gutiérrez, a researcher and surgeon at the University of Pittsburgh in Pennsylvania. “Besides the dream of making organs for transplantation, there are a lot of things we can learn from these systems,” he says — including a better basic understanding of cell organization in the heart and new ideas about how to fix one.
The idea of bioengineering a heart is a huge ambition that Doris Taylor has, which has the potential to save thousands of lives should it work. However the point made by Alejandro Soto-Gutiérrez, I feel is extremely valid. The research into bioengineering is worth while, because not only has this worked with tubular structures, even if it doesn't work with the more complex structures such as the heart. It will educate surgeons and scientists further which may help inspire new methods of surgical options or even medication.
http://www.nature.com/news/tissue-engineering-how-to-build-a-heart-1.13327
Holly
Taylor is in the vanguard of researchers looking to engineer entire new organs, to enable transplants without the risk of rejection by the recipient's immune system. The strategy is simple enough in principle. First remove all the cells from a dead organ — it does not even have to be from a human — then take the protein scaffold left behind and repopulate it with stem cells immunologically matched to the patient in need. Voilà! The crippling shortage of transplantable organs around the world is solved.
In practice, however, the process is beset with tremendous challenges. Researchers have had some success with growing and transplanting hollow, relatively simple organs such as tracheas and bladders (seego.nature.com/zvuxed). But growing solid organs such as kidneys or lungs means getting dozens of cell types into exactly the right positions, and simultaneously growing complete networks of blood vessels to keep them alive. The new organs must be sterile, able to grow if the patient is young, and at least nominally able to repair themselves. Most importantly, they have to work — ideally, for a lifetime. The heart is the third most needed organ after the kidney and the liver, with a waiting list of about 3,500 in the United States alone, but it poses extra challenges for transplantation and bioengineering. The heart must beat constantly to pump some 7,000 litres of blood per day without a back-up. It has chambers and valves constructed from several different types of specialized muscle cells called cardiomyocytes. And donor hearts are rare, because they are often damaged by disease or resuscitation efforts, so a steady supply of bioengineered organs would be welcome.
Taylor, who led some of the first successful experiments to build rat hearts1, is optimistic about this ultimate challenge in tissue engineering. “I think it's eminently doable,” she says, quickly adding, “I don't think it's simple.” Some colleagues are less optimistic. Paolo Macchiarini, a thoracic surgeon and scientist at the Karolinska Institute in Stockholm, who has transplanted bioengineered tracheas into several patients, says that although tissue engineering could become routine for replacing tubular structures such as windpipes, arteries and oesophagi, he is “not confident that this will happen with more complex organs”.
Yet the effort may be worthwhile even if it fails, says Alejandro Soto-Gutiérrez, a researcher and surgeon at the University of Pittsburgh in Pennsylvania. “Besides the dream of making organs for transplantation, there are a lot of things we can learn from these systems,” he says — including a better basic understanding of cell organization in the heart and new ideas about how to fix one.
The idea of bioengineering a heart is a huge ambition that Doris Taylor has, which has the potential to save thousands of lives should it work. However the point made by Alejandro Soto-Gutiérrez, I feel is extremely valid. The research into bioengineering is worth while, because not only has this worked with tubular structures, even if it doesn't work with the more complex structures such as the heart. It will educate surgeons and scientists further which may help inspire new methods of surgical options or even medication.
http://www.nature.com/news/tissue-engineering-how-to-build-a-heart-1.13327
Holly
|
http://read.feedly.com/html?url=http://www.wired.com/wiredscience/2014/02/beautiful-fish-skeletons/&theme=white
Dead fish can be surprisingly beautiful. Hiding beneath their scales is an elegant, skeletal architecture, a complex biological scaffold that with some skill, can become art. Adam Summers, who studies vertebrate biomechanics at the University of Washington, takes photos of the insides of fish as part of his research. But the process is a lot more involved than snapping a pic with a cellphone. His pictures can take days to produce, and they reveal the intricate bony or cartilaginous infrastructure inside marine creatures like rays, skates, and fish. Summers’ work is now featured in an exhibit at the Seattle Aquarium that pairs each photograph with a poem. Summers follows a standard, decades-old procedure to visualize the different types of tissue. First, you start with your organism – say, a dead fish. Next, you follow a few fixation steps that both preserve tissues and make them more permeable to dyes and enzymes. Then it’s time to color the different tissues. Summers uses Alcian Blue to stain cartilage, and Alizarin Red S to dye the bony bits. Next, you clear away the fish’s flesh with a digestive enzyme called trypsin (trypsin doesn’t attack collagen, the fiber that holds skin and bone together), and bleach out dark pigments with hydrogen peroxide. Lastly, you submerge the now-colorful organism in glycerin, a clear and colorless solution that renders the fish transparent except for the colored bits. Then you take its picture. Summers uses a macro lens on a DSLR and places the submerged specimens on a color-corrected LED table. The results, as you can see, are exquisite. -Jed |
The story of decellularisation
This video shows the evolution of decellularisation from the beginning of cloning to organ regrowth. It explains the processes involved and the series of events which lead to the discovery of decellularisation.
Thulangana Ganeshan.
Thulangana Ganeshan.